
Lot tracking and batch traceability software
Trace every ingredient from supplier to customer. Katana’s end-to-end lot tracking tells you exactly which batches went into which products and which customers received them — so a recall investigation takes minutes, not days.
Why regulated manufacturers need lot tracking software
Lot tracking software assigns unique identifiers to each batch of materials received and products manufactured, creating a complete chain of traceability. For food, cosmetics, and supplement manufacturers, this means you can trace any ingredient from the supplier lot through production batches to the specific customers who received the finished product meeting FDA and regulatory requirements while turning recall investigations from days-long audits into minutes-long queries.
Supplier-to-customer traceability
Know which supplier lot went into which production batch and which customer received the finished product. The complete chain, recorded automatically.
Instant stock transfers
If an ingredient is recalled, instantly identify every affected production batch and every customer who received it. No spreadsheet searching, no warehouse floor audits.
Expiry date management
Track best-before dates at the lot level. Enforce FEFO picking to use oldest stock first. Get alerts before materials expire so nothing goes to waste or into production.
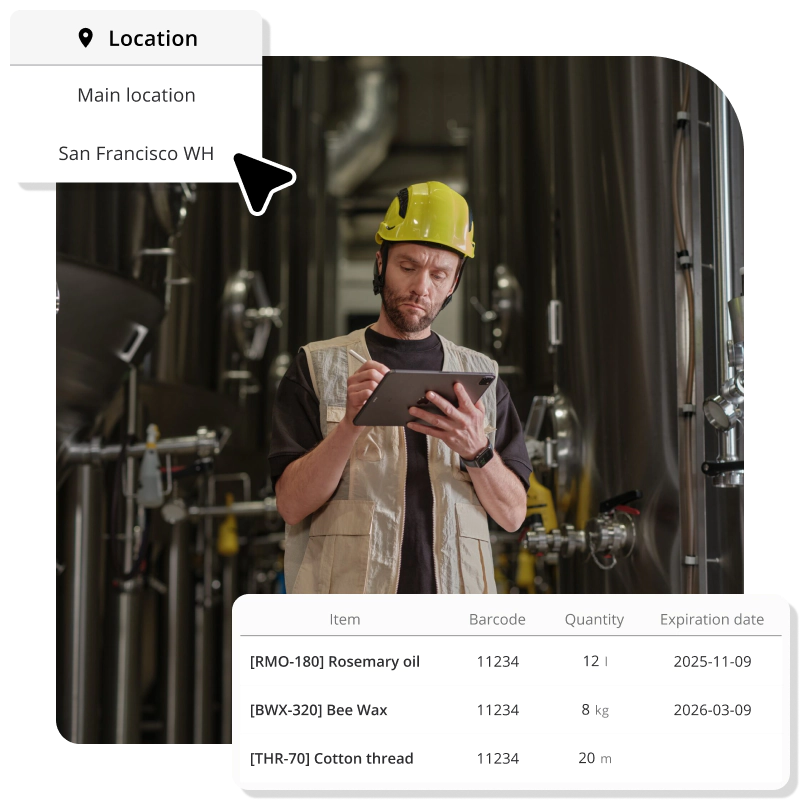
End-to-end traceability: from receiving dock to customer door
When a shipment of ingredients arrives, you assign a lot number in Katana. When those ingredients go into production, the lot numbers are linked to the finished product batch. When that batch ships to customers, the link extends to the sales order.
If anything goes wrong at any point in the chain, such as a contaminated ingredient, quality defect, or mislabeling, you can trace forward or backward through the entire history in seconds.
“Katana is the only one that offers end-to-end full traceability. The health inspector would demand it.”
— Food & beverage manufacturer

Handle recalls without panic
A supplier calls at 3 PM: lot #FL-2024-089 of flour has been flagged for contamination. What happens next?
Without lot tracking: Warehouse team spends days checking paper records. Production stops. Customers are contacted in batches over a week. You hope you caught everything.
With Katana: Search lot #FL-2024-089. Instantly see every production batch that used it. See every customer who received those batches. Generate a recall contact list in minutes. Document everything for the FDA
“If that’s being recalled, it would easily tell us who we need to contact.”
— Katana customer on recall readiness
Expiry date tracking and FEFO enforcement
Track best-before and use-by dates at the lot level for every raw material and finished good. Katana enforces First Expiry, First Out picking so your warehouse team always uses the oldest stock first.
- Set expiry dates when receiving material lots
- Visual alerts when materials approach or pass expiry
- FEFO picking suggestions in production and fulfillment
- Prevent expired materials from being used in production
Is your business recall-ready?
If a health inspector or the FDA asked you to trace an ingredient through your entire production chain today, how long would it take? With Katana, the answer is minutes.
Industries that need lot tracking
Explore Katana’s inventory capabilities
Frequently asked questions
Lot tracking assigns a unique identifier to each batch of materials received or products manufactured. This allows manufacturers to trace every ingredient from the supplier through production to the end customer. If a quality issue arises, you can identify exactly which products are affected and which customers received them — essential for regulated industries like food, cosmetics, and supplements.
Batch traceability for food manufacturers tracks every ingredient lot through the production process. When flour lot #A123 goes into bread batch #B456, that link is recorded. If flour lot #A123 is later recalled, you can instantly identify all bread batches that used it and all customers who received those batches — turning a days-long investigation into a minutes-long query.
Yes. The FDA requires food manufacturers to maintain records that identify the immediate previous source and immediate subsequent recipient of food (the ‘one-up, one-back’ rule under the Bioterrorism Act). Lot tracking software automates this compliance by recording supplier lot numbers at receiving, linking them through production, and tracking which customers received each finished batch.
Yes. Katana tracks expiry dates at the lot level for both raw materials and finished goods. You can enforce FEFO (First Expiry, First Out) picking, get alerts before materials expire, and ensure that expired ingredients never enter production.
Full traceability, from supplier to customer
Start tracking lots and batches with Katana’s free plan. No credit card, no per-user fees. Set up lot tracking for your materials and products today.
